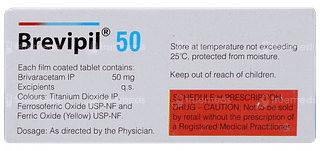
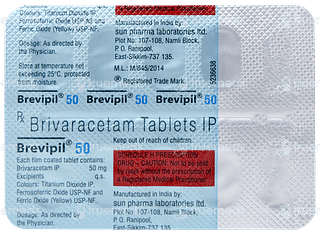
Brevipil 50 Tablet is an anti-epileptic medication used to treat seizures in patients with epilepsy, especially when combined with other anti-seizure medications. It contains Brivaracetam, which stabilizes abnormal electrical activity in the brain, reducing the frequency and severity of seizures. Brevipil is effective in managing seizures that are not fully controlled by other medications, offering additional support for those with epilepsy.
As with any medication, it is crucial to consider potential side effects such as drowsiness, dizziness, nausea, and headache. However, it is worth noting that Brevipil generally exhibits good tolerability and can be well-managed with appropriate monitoring.
Close collaboration between patients, caregivers, and healthcare providers is crucial to ensure optimal dosing, monitor treatment response, and address any concerns or questions that may arise during therapy.